Color, Chemistry, and Craft: Investigating the Effects of pH and Water Hardness on Coconut-Husk Natural Dyeing
Chinmay Kumar
Fall 2025
HIST GU4962: Making and Knowing in Early Modern Europe: Hands-On History
Abstract
This project examines how pH and water hardness influence the tone and intensity of color produced from a natural coconut-husk dye. Unlike many natural dye experiments, no mordant was used, allowing the effects of acidity, alkalinity, and magnesium-induced hardness to be observed in isolation. Across six controlled dye baths (acidic, neutral, and basic x soft and hard water), hard-water samples universally produced deeper tones, likely due to magnesium ions acting as quasi-mordants and enhancing tannin–fiber interactions. Acidic conditions further darkened color, while basic conditions yielded lighter, dustier hues. The experiment is interpreted through early modern frameworks of sensory empiricism, demonstrating how artisans historically relied on perception rather than instrumentation to navigate similar chemical variability.
Introduction
In early modern Europe, dyeing was a chemical art practiced before chemistry as a formal discipline. Artisans relied on sight, touch, temperature, smell, and local knowledge rather than standardized reagents or analytical tools. Their practices were grounded in embodied knowledge: repeated, sensory engagement with materials constituted a legitimate mode of knowing. Dyers learned not through abstract theory but through experimentation, through watching colors bloom or fail, feeling fibers stiffen or soften, and noting how seemingly minor variations in water or heat altered results. Professor’s frequent invocation of “process over product” captures this epistemology well, foregrounding making as a way of understanding material behavior rather than merely producing a finished object.
This capstone reconstructs such a process by investigating the dyeing properties of coconut husk, a tannin-rich material widely used across tropical regions yet under-represented in European dye treatises. Chemically, tannins are a diverse class of high–molecular-weight polyphenolic compounds produced by plants as defensive metabolites. They are characterized by multiple phenolic hydroxyl (–OH) groups attached to aromatic rings, a structure that allows them to readily form hydrogen bonds and metal–ligand complexes. Two major categories are typically distinguished: hydrolyzable tannins, composed of gallic or ellagic acid units esterified to a sugar core, and condensed tannins (proanthocyanidins), polymeric flavonoid structures that are more resistant to hydrolysis. Coconut husk is particularly rich in condensed tannins, which are known for their strong affinity for cellulose fibers and their pronounced color shifts in the presence of metal ions.
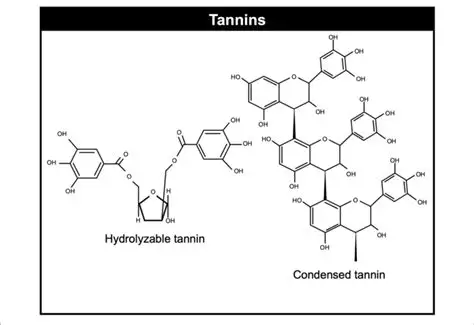
Fig 1: Tannin Structure as taken From Ku-Vera et al., 2020
The dyeing action of tannins arises from these chemical properties. Phenolic groups can bind directly to cellulose through hydrogen bonding, while also chelating (coordinating with) dissolved metal ions such as calcium, magnesium, or iron. These interactions influence both color intensity and hue, producing browns, reds, or grays depending on environmental conditions. In the absence of a separate mordant, tannins themselves function as reactive agents, forming insoluble complexes within the fiber matrix. This dual behavior acting simultaneously as dye and mordant made tannin-rich materials especially valuable in historical dyeing, even if their chemical mechanisms were not explicitly articulated.
Crucially, these interactions are highly sensitive to pH and water composition. Acidity affects the ionization state of phenolic groups, altering their binding capacity, while dissolved minerals can dramatically shift chromatic outcomes. Early modern dyers were acutely aware of these effects, even if they framed them empirically rather than theoretically. Water was never chemically neutral in premodern dyeing: its mineral content varied widely by geography, depending on local geology and water sources. “Hard” waters, rich in calcium and magnesium carbonates, were common in regions with limestone bedrock, while “soft” waters predominated in areas supplied by rainwater, peat, or granitic terrain. Dyers routinely remarked that certain towns produced superior colors because of their water, and recipes often specified the use of river, rain, or well water without further explanation. Early modern technical literature and workshop practice consistently attest to this sensitivity. Artisans knew that some waters “dulled” colors or caused them to shift toward gray or brown, while others allowed brighter or more stable hues.
Iron contamination, sometimes introduced unintentionally through pipes, vats, or tools, was notorious for darkening tannin dyes, a phenomenon now understood as the formation of iron–tannin complexes. Although terms like “hardness” or “ionic concentration” were absent, dyers managed these variables through pragmatic strategies: settling water, boiling it, choosing specific sources, or adjusting processes through repeated trials. Water, like heat or time, was treated as an active ingredient rather than an inert solvent.
By manipulating pH (acidic, neutral, basic) and hardness (soft water versus magnesium-rich Epsom-salt solutions), this project recreates a subset of the chemical conditions early modern dyers encountered implicitly through their local environments. Cotton, a cellulose-based fiber increasingly present in early modern global trade, provides a consistent substrate through which these interactions can be observed. Because no external mordant was employed, all color variation arises from tannins’ intrinsic chemistry and their interactions with dissolved minerals: precisely the kind of material contingency that early modern artisans navigated through embodied experimentation rather than abstract chemical theory. In this way, the project not only examines coconut husk as a dye material but also reenacts a historically grounded mode of knowing through making, where chemical phenomena reveal themselves through process.
Research Questions and Hypotheses
Primary Research Questions:
How does pH influence the color intensity and tone of coconut-husk dye?
How does water hardness affect dye uptake and color saturation?
Secondary Research Questions:
How would early modern artisans have observed such chemical variability?
How can modern reconstruction help re-experience that sensory-based epistemology?
Hypotheses:
Acidic baths will intensify color because tannins bind more effectively at low pH.
Hard water containing magnesium will act as a quasi-mordant, deepening tone.
Basic baths will yield paler tones due to reduced protonation of tannins.
No expectations concerning protein vs. cellulose fibers were formed for this experiment because only cotton handkerchiefs were dyed.
Methods
Table 1: Conditions for Experimental Setup
| Condition | Modifier | Water Type | Notation |
|---|---|---|---|
| Acidic Soft | 1 g Vinegar | Tap | A |
| Acidic Hard | 1 g Vinegar + 1 g Epsom | Tap | AH |
| Neutral Soft | \ | Tap | N |
| Neutral Hard | 1 g Epsom | Tap | NH |
| Basic Soft | 1 g Baking Soda | Tap | B |
| Basic Hard | 1 g Baking Soda + 1 g Epsom | Tap | BH |

Fig 2: Experimental Setup Prior to Cotton Handkerchief Immersion
Table 2: Orientation from Top Right to Bottom Left:
| Acidic Soft | Acidic Hard |
|---|---|
| Basic Soft | Basic Hard |
| Neutral Soft | Neutral |
General Procedure:
Division: 40 mL of tap water poured into each 100 mL beaker, plus 10 mL of the corresponding modifier solution.
Simmering: Coconut husk simmered in 50 mL solution for ~15 minutes.
Dyeing: Cotton swatches were submerged, heated, stirred for 15 minutes, then cooled and patted and aired to dry for ~5 minutes.
Rinsing: Each swatch was rinsed with distilled water, dried, and labeled.
Observations Taken from Lab Notebook
Quantities and Chemical Behavior
40 mL water + 10 mL modifier = 50 mL total bath volume should have been homogenous but different surface area of exposed solute caused apparent different volume levels.
1 g husk added to each bath; solids not strained may have led to uneven exposure of cotton handkerchief to tannins.
Hard neutral (N–H) reached a rolling boil first, consistent with colligative effects (dissolved ions lowering vapor pressure and disrupting intermolecular forces).
Baking soda reacted immediately upon heating (CO₂ release).
Some fluid evaporation caused slightly shortened simmering time.
Concern for potential side reactions: the epsom salt (magnesium sulfate) and vinegar (acetic acid)/baking soda (sodium bicarbonate) may have potentially reacted leading to the insignificant observable difference between acid + base/ hard group.
Sensory Notes
Acidic baths smelled sharper and more in line with my past experience with flavonoids in Organic Chemistry Lab.
Basic baths smelled soapy, like laundry during a warm rinse for soft clothing.
Hard-water baths were slightly more difficult to stir and the texture felt a little rougher or potentially more granular when removing the cotton handkerchief from the simmering husk bath.
The process felt both “clinical and uniform” yet “homey and warm,” reflecting the fusion of measurement and tactility.
Results
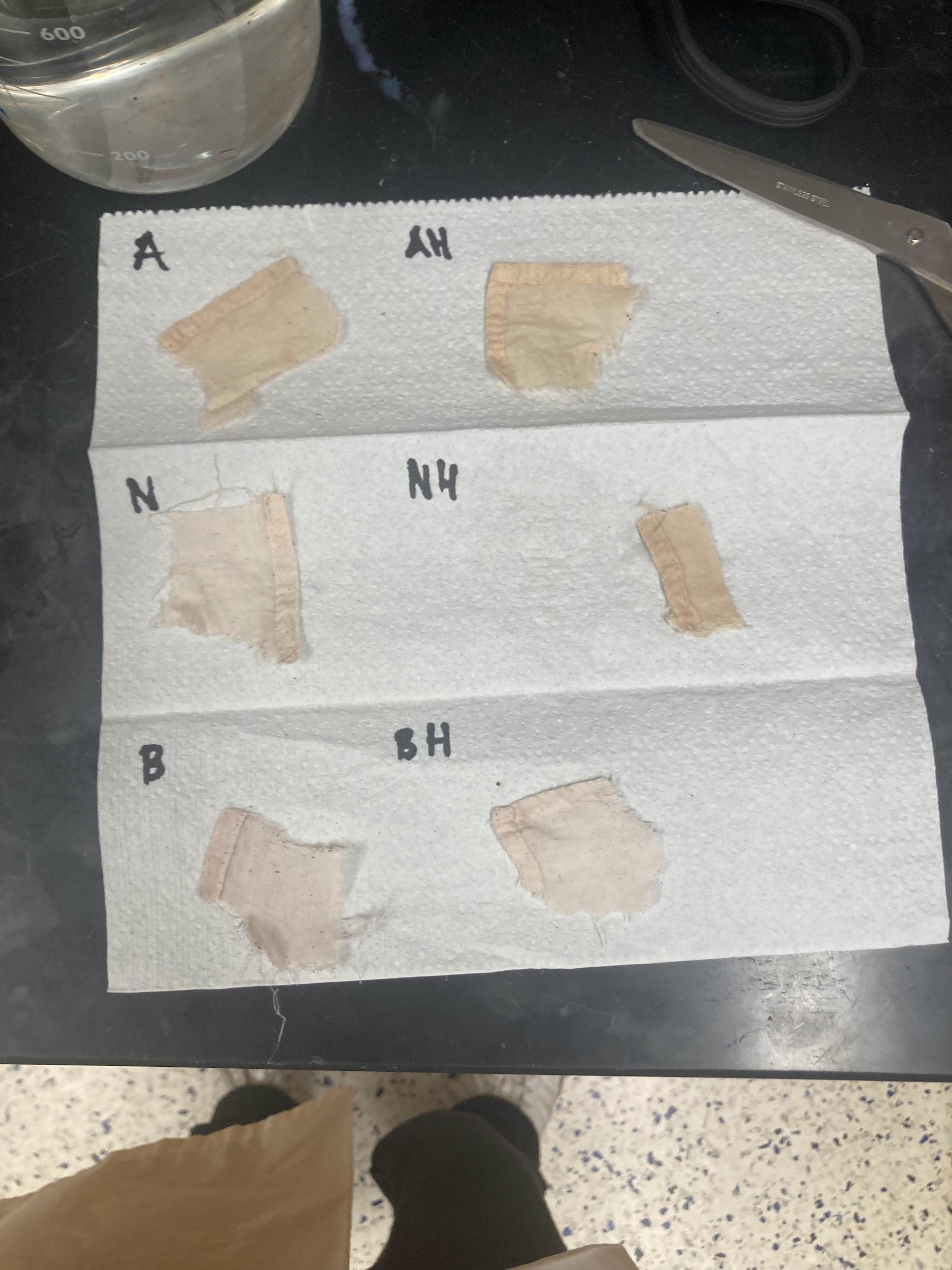
Figure 3: Visual Outcomes Immediately After Removal from Dye Bath
Table 3: Visual Outcome Descriptions
| Condition | Color Description | Notes |
|---|---|---|
| A - Acid Soft | Muted golden beige | Warm undertones |
| A - Acid Hard | Similar muted golden-brown beige | A slight stretch to say cool undertones but certainly a slightly darker complexion on the cloth |
| N - Neutral | Pale reddish brown | Very cool undertones developing from the contrast between the white handkerchief lightly dyed without mordant and the leftover detritus caked onto the cloth |
| NH - Neutral Hard | Darker warmer yellowish brown | Warmer undertones, noticeably darker than the neutral-soft condition |
| B - Basic Soft | Pale reddish brown | Relatively cool undertones |
| BH - Basic Hard | Paler reddish brown with a slight graying | Again cool undertones |
Reflection
This project demonstrates that water chemistry alone, specifically pH and mineral hardness, can produce substantial and reproducible differences in tannin-based dyeing, even in the absence of an external mordant. Across all trials, harder water consistently yielded darker tones than soft water, while acidic conditions generally produced stronger color uptake than neutral or basic baths. Basic environments tended toward weaker, more washed-out hues. However, the anticipated sharp dichotomy between acidic and basic conditions did not always manifest, and the acid-hard/acid and base-hard/base dye baths exhibited intermediate or overlapping color intensities. Rather than indicating experimental failure, this outcome reflects the chemical and material complexity of tannin dye systems and points to meaningful sources of experimental uncertainty.
From a chemical perspective, magnesium ions in Epsom salt appear to function as a quasi-mordant, coordinating with tannins and strengthening their attachment to cellulose fibers. Acidic conditions promote protonation of phenolic groups, which typically enhances hydrogen bonding and fiber affinity, while basic conditions deprotonate these groups and weaken binding. Yet in practice, these effects did not always resolve into a simple acidic–dark versus basic–light contrast. One source of ambiguity likely arises from side reactions within the dye bath itself. Coconut husk contains not only tannins but also lignin fragments, sugars, and organic acids, which may participate in secondary reactions under heat. In acidic environments, partial hydrolysis of tannin polymers could increase the number of reactive sites, while in basic environments, oxidative degradation or solubilization may alter both tannin structure and effective concentration. These processes may have blurred the expected pH-dependent trends.
Additional sources of experimental error further complicate interpretation. pH levels, though initially adjusted for with appropriate additions, may have shifted during heating due to volatilization of acetic acid, buffering effects from the plant material, or interactions with dissolved salts. Localized pH gradients within the dye bath, uneven agitation, or variable fiber exposure could also have contributed to nonuniform uptake. Magnesium ions may have altered ionic strength in ways that affected tannin aggregation rather than simply enhancing binding, producing nonlinear color responses. These factors suggest that the observed color outcomes reflect a dynamic chemical environment rather than a static set of controlled conditions.
The unexpected result that the neutral hard condition produced the richest hue underscores this point. Rather than contradicting the underlying chemistry, this outcome suggests that intermediate pH conditions may optimize the balance between tannin solubility, mineral coordination, and fiber accessibility. Such nonlinear behavior would have been familiar to early modern dyers, for whom small, often imperceptible variations in water or heat could yield dramatically different results. In this sense, the absence of a clean acidic/basic dichotomy is itself historically informative, mirroring the unpredictability that made dyeing a skill acquired through experience rather than formula.
Conducting this experiment made clear how much dyeing depends on embodied judgment. Color assessment occurred continuously, not solely at the endpoint: by watching the bath darken or stall, noticing shifts in smell or surface activity, feeling changes in fabric texture, and attending to bubbling or cloudiness that suggested ongoing reactions. These sensory observations often revealed when the bath was behaving “unexpectedly,” prompting interpretation rather than correction. This mode of attention echoes the ethos of the Making and Knowing Project, in which historical understanding arises through sustained engagement with materials and their resistances rather than through idealized control.
Removing the mordant simplified the experimental design but sharpened awareness of uncertainty. By limiting the system to plant material, water, and heat, the experiment foregrounded how easily side reactions and environmental fluctuations can disrupt theoretical expectations. Future extensions could address these uncertainties by monitoring pH continuously throughout heating, isolating tannin fractions prior to dyeing, or introducing controlled trace metals to disentangle their effects from pH-driven changes. Yet even these refinements would not eliminate such contingency; they would merely shift its form.
Ultimately, watching six nearly identical dye baths resolve into six outcomes, without a perfectly predictable acidic/basic divide, made tangible the sensitivity of natural dyes to environmental conditions and the interpretive labor required to work with them. What began as a chemical investigation evolved into an exercise in historical empathy as I began to understand a smidge of the frustration the early modern dyers may have felt when attempting to reproduce a color they had once obtained. By confronting ambiguity, side reactions, and experimental error, this project reenacts the epistemic conditions of early modern dyeing, demonstrating the inseparability of craft, chemistry, and environment and fulfilling the central aim of Making and Knowing: a fusion of intellectual inquiry with embodied, material practice through which historical understanding is not merely described, but enacted.
Bibliography
Autumn Color Co., Ltd. “The Importance of pH Control in Basic Dyeing.” 2025. https://autumnchem.com/the-importance-of-ph-control-in-basic-dyeing/
Benli, H. “Bio-mordants: A Review.” PubMed Central (PMC), 2024.
Braide, O. O., Labode, O. J., Amuda, M. O., and Bolarin, O. O. “Extraction of Dye from Coconut Husk (Cocos nucifera) and Its Effect on Cotton Fabric.” European Journal of Theoretical and Applied Sciences 3, no. 2 (2025).
Burch, P. “Dyeing / FAQ / pH.” P. Burch Dyeing FAQ. http://www.pburch.net/dyeing/FAQ/ph.shtml
Ku-Vera, Frontiers of Veterinary Science. “Role of Secondary Plant Metabolites on Enteric Methane Mitigation in Ruminants.” Frontiers in Veterinary Science (August 2020). https://doi.org/10.3389/fvets.2020.00584.
“How to Adjust the pH Value During the Dyeing Process on the HTHP Sample Dyeing M-36339.” HZDMCN Blog. https://www.hzdmcn.com/blog/how-to-adjust-the-ph-value-during-the-dyeing-process-on-the-hthp-sample-dyeing-m-36339.html
Live to Plant. “The Role of pH in Dyeing: How It Affects Colors.” 2025. https://livetoplant.com/the-role-of-ph-in-dyeing-how-it-affects-colors/
Sirisangsawang, R., et al. “Optimization of Tannin Extraction from Coconut Coir.” PubMed Central (PMC), 2023.
Uddin, M. G. “Effects of Different Mordants on Silk Fabric Dyed with …” 2014. Wiley Online Library.
Wilson, H., et al. “Production and Validation of Model Iron-Tannate Dyed Textiles.” BMC Chemistry (2012).